|
The trends in ionisation energies across periods and down groups can be explained in terms of the atomic size, nuclear charge and the screening effect due to inner shell electrons.C4.1b explain how observed simple properties of Groups 1, 7 and 0 depend on the outer shell of electrons of the atoms and predict properties from given trends down the groups. 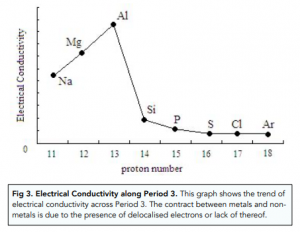
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
